Aim
To study quantity of casein in different samples of milk.
Theory
Milk contains 3 to 4% casein suspended in water in the colloidal form. It is precipitated in a weakly acidic medium.
Apparatus Required
Funnel, funnel stand , glass rod , filter paper, weight box , test tubes, pestle and mortar.
Chemicals Required –
(i) Different samples of milk.
(ii) Saturated ammonium sulphate solution.
(iii) 1 % acetic acid solution
Procedure –
1. Wash the beaker (250 ml) with the distilled water and dry it.
2. Take 20 ml of buffalo’s milk in 250 ml beaker and find its weight.
3. Add 20 ml saturated solution of ammonium sulphate slowly with stirring. Fat and casein will separate out as precipitate.
4. Filter the above solution and transfer the precipitate in another beaker.
5. Treat the above precipitate with 30 ml distilled water. Casein dissolves forming milky solution whereas fat remains as such.
6. Warm the above contents of the beaker to 40 – 45°C on a low flame. Now, add 1% acetic acid solution drop wise with stirring when casein gets precipitated.
7. Filter the precipitated casein and wash with distilled water and dry it.
8. Find the weight of dry precipitate.
9. Repeat the whole experiment with cow’s milk, goat’s milk and sheep’s milk.
Observations
Volume of milk taken in each case = 20 ml Weight of milk taken = W₁ g Weight of Casein isolated = W₂ g Percentage of casein = (Weight of Casein x 100)/ Weight of milk
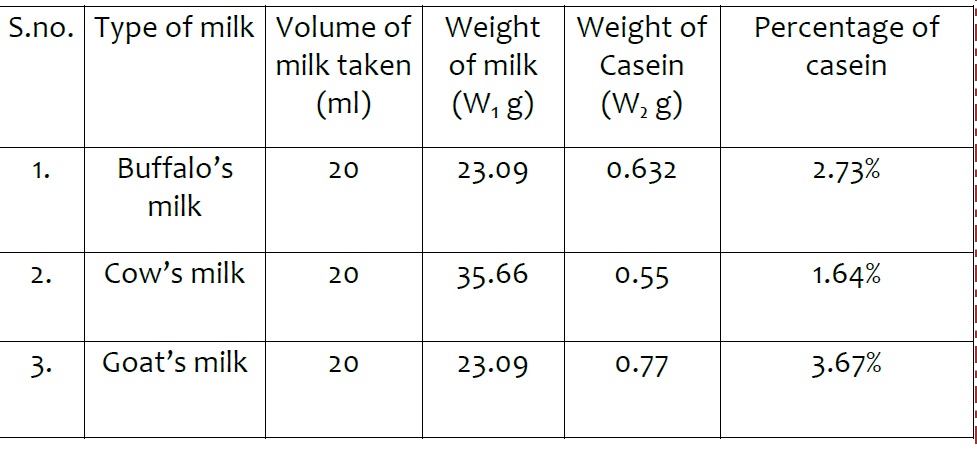
Result – Different Samples of milk contains different percentage of casein. Highest percentage of casein is present in Goat’s milk.
Precautions –
1. Handle apparatus and chemicals carefully.
2. Add ammonium sulphate solution very slowly.
3. Stir milk while adding chemicals.
4. Do not disturb milk after adding ammonium sulphate solution and wait some time for fat and casein to precipitate out.
5. Take the amount readings carefully with digital weighing machine only.
